Research
At the Center of Cancer Systems Biology I study the systemic response of a host to the presence of cancer. The immune system is my major focus in this endeavor. I consider both the tumor-inhibiting and the tumor-promoting actions of bone marrow derived cells in tumor development as well as the modulation of these dynamics through aging, anti-cancer therapies, and low dose radiation exposure.
Tumor-Promoting Inflammation
The presence of cancer within a host initiates a systemic immune response towards the transformed cells. Inflammatory immune cells such as neutrophils, platelets, macrophages, and natural killer cells, are recruited to the tumor site where they initiate the wound healing process. Tumors, sometimes viewed as wounds that never heal, can be promoted by these inflammatory actions. Once the adaptive immune response is activated by dendritic cells and macrophages, CD8+ T cells, or cytotoxic T lymphocytes, infiltrate the tumor and induce apoptosis in the target tumor cells.
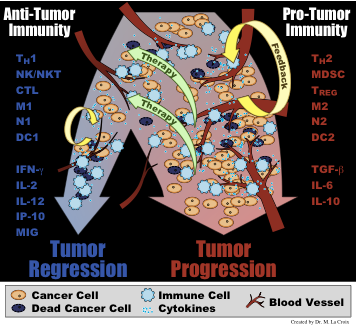
Depending on the cytokines and other signals present in the tumor microenvironment, recruited immune cells will either form a pro-tumor immunity (typified by cytokines such as TGF-β, IL-6, and IL-10 and cells such as M2 macrophages, Th-2 T helper cells, and myeloid derived suppressor cells) or an anti-tumor immunity (typified by cytokines such as IFN-γ, IL-2, and IL-12 and cells such as M1 macrophages, Th-1 T helper cells, and cytotoxic lymphocytes).
To investigate the role of tumor-promoting inflammation, an emerging hallmark of cancer, I, together with my postdoctoral supervisor Dr. Philip Hahnfeldt, have developed a mathematical model for cancer-immune interactions that can capture both the pro-angiogenic, tumor-progressing actions of a pro-tumor inflammatory microenvironment, and the anti-angiogenic, tumor-inhibiting actions of an anti-tumor inflammatory microenvironment. This model utilizes principles of generalized logistic growth, which captures some of the inherent variability underlying tumor growth in an immune competent host that is often neglected in macroscopic measurements and in mathematical models. From model simulations, the two types of inflammation (pro-tumor or anti-tumor) resolve into two fundamentally different classes of outcomes, where inflammation-enhanced tumor progression must either result in a decreased tumor burden, as in the anti-tumor case, or in an increased tumor burden, as in the pro-tumor case. These results suggest that, in some cases, fast tumor growth may be advantageous, if it leads to a significantly smaller tumor burden. In such cases, it is possible that treatments should be targeted towards enhancing the stability of an anti-tumor inflammatory environment instead of towards immediate tumor regression.
Immunoediting and the Emergence of Immune Resistance
The immune response poses a second barrier to tumor growth after the angiogenic switch. Immune surveillance of tissues allows for early detection of transformed cells. If these transformed cells are not recognized as “self”, they are eliminated by the immune system. Through repeated exposure of the transformed cells to this immune selection process, various phenotypes can arise within the cancer cell population, creating a heterogeneous population of neoplastic cells. These immunoedited cells may develop the ability to evade the immune response and grow in an uncontrolled manner.

After prolonged periods of immune-induced tumor dormancy, T cells can lose effectiveness in their cytotoxicity. This loss may be due to either T cell tolerization to the cancer cells or to an increased cancer cell resistance to immune attack. Both of these mechanisms are intertwined in the process of immunoediting that can lead to tumor escape from immune control.
Brain Biomechanics
My doctoral research was in the area of brain biomechanics, or more specifically, the study of how hydrocephalus develops in the brain. My PhD supervisor was Professor Siv Sivaloganathan at UW, and a copy of my thesis can be found here. Other areas of interest include continuum mechanics, porous media, multiscale modelling, population dynamics, and imaging science.
My masters thesis, under the supervision of Professor Ed Vrscay at UW was on mutual information based methods to localize image registration with specific applications to medical imaging. A copy of my masters thesis can be found here.
News
This summer I'll be a mentor for the ICBP's summer undergraduate research program fellow at our CCSB.
Recent Publication
Here's an upcoming review article on the systemic nature of cancer and mathematical approaches that encompass this aspect of the disease, in Mathematical Oncology [Eds. A d'Onofrio and A Gandolfi], June 22, 2014.